 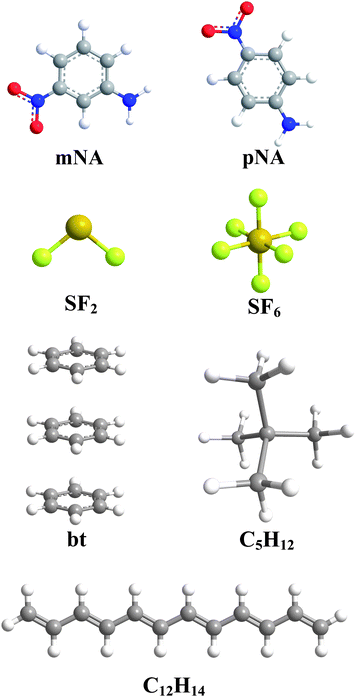 
In 1984, large quantities of Sevin were accidentally released in Bhopal, India, when water leaked into storage tanks. We will demonstrate with methyl isocyanate (CH 3–N=C=O), a volatile and highly toxic molecule that is used to produce the pesticide Sevin. The VSEPR model can be used to predict the structure of somewhat more complex molecules with no single central atom by treating them as linked AX mE n fragments. With no lone pair repulsions, we do not expect any bond angles to deviate from the ideal.ĭ The PF 5 molecule has five nuclei and no lone pairs of electrons, so its molecular geometry is trigonal bipyramidal. 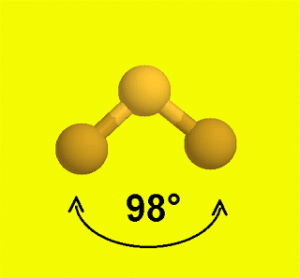
Notice that this gives a total of five electron pairs. Figure \(\PageIndex\)): (CC BY-NC-SA anonymous)Ĭ All electron groups are bonding pairs, so PF 5 is designated as AX 5. This theory is very simplistic and does not account for the subtleties of orbital interactions that influence molecular shapes however, the simple VSEPR counting procedure accurately predicts the three-dimensional structures of a large number of compounds, which cannot be predicted using the Lewis electron-pair approach. The premise of the VSEPR theory is that electron pairs located in bonds and lone pairs repel each other and will therefore adopt the geometry that places electron pairs as far apart from each other as possible. The VSEPR model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a central metal atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
